Product
MyopiaX®

Each session with MyopiaX delivers selective ocular light stimulation through VR game. Use the MyopiaX® to monitor session completion and performance to optimize treatment compliance. Learn more about how MyopiaX® works on our Science page.


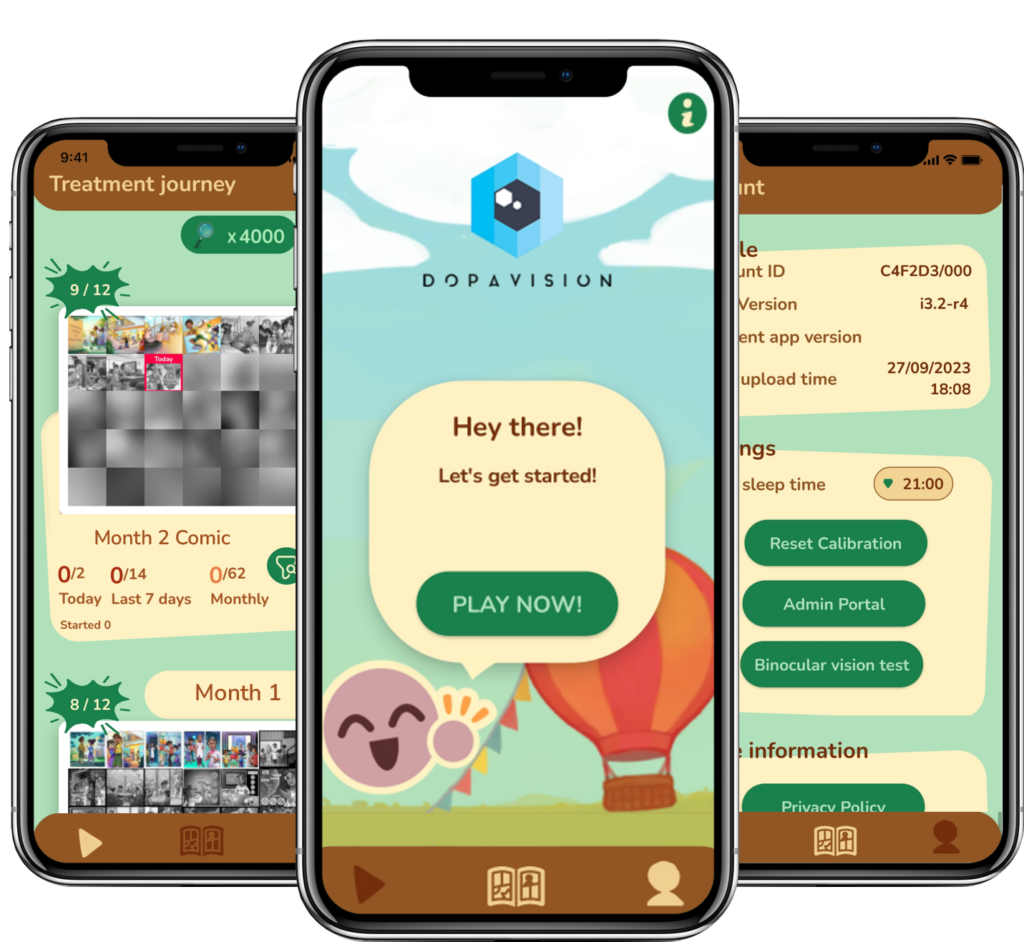
Our App
MyopiaX® is designed for everyday life, leveraging consumer technology to administer selective ocular light stimulation through an interactive digital experience. Developed to deliver an active treatment, MyopiaX® adheres to medical device regulations. The MyopiaX®’s intuitive app interface makes at-home use simple, transforming twice-daily sessions into a child-friendly, therapeutic routine.
Child-Friendly Alternative


Developed According to Medical Device Standards
MyopiaX® is developed to meet medical device safety standards for everyday use, backed by comprehensive technical documentation covering all essential norms, including software lifecycle management (EN 62304) and risk management (ISO 14971). Our goal is to achieve certification as a medical device under European and international regulations. Results from ongoing research and clinical trials will support regulatory activities to certify MyopiaX® as a safe, effective treatment for myopia control.

More About MyopiaX®
MyopiaX® is designed to be used daily. Each session lasts for several minutes and delivers selective ocular light stimulation while users interact with virtual reality content. The virtual reality content includes mini-games, a customizable avatar, and interactive quests. A variety of fun and educational content means that MyopiaX® is always engaging for users.



